|
The hot gasses (in the form of steam) have to release energy into the environment in the form of heat to cool to the point that they can form liquid water, meaning that the formation of H 2O is exothermic. H represents enthalpy, which is the sum of a systems internal energy + the product of the systems pressure and volume. This makes sense - H 2 and O 2 are gasses, while H 2O, the product, is a liquid. Since the sign is negative, we know that our reaction is exothermic. Reaction thermochemistry data for over 8000 reactions. Phase transition enthalpies and temperatures. In our example, our final answer is -13608 J. The NIST Chemistry WebBook contains: Thermochemical data for over 7000 organic and small inorganic compounds: Enthalpy of formation. There is a 'heat tax' for every energy transaction Which of the following processes have a delta S>0 A) CH3OH (l) -> CH3OH (s) B) N2 (g) + 3H2 (g) -> 2NH3 (g) C) CH4 (g) + H20 (g) -> CO (g) + 3H2 (g) D) NA2CO3 (s) + H20 (g) + CO2 -> 2NaHCO3 (s) E) All of the above C.Beware strongly exothermic reactions - these can sometimes signify a large release of energy, which, if rapid enough, can cause an explosion. The larger the number itself is, the more exo- or endo- thermic the reaction is. Knowing that the specific heat of water is 4.184 J g-1 C o, calculate the average H sub /g CO 2 and the average H sub /mol CO 2. On the other hand, if the sign is negative, the reaction is exothermic. Calculations: The heat gained by the CO 2 ( H sub) is equal to the heat released by the water. If the sign of your final answer for ∆H is positive, the reaction is endothermic. One of the most common reasons that ∆H is calculated for various reactions is to determine whether the reaction is exothermic (loses energy and gives off heat) or endothermic (gains energy and absorbs heat). 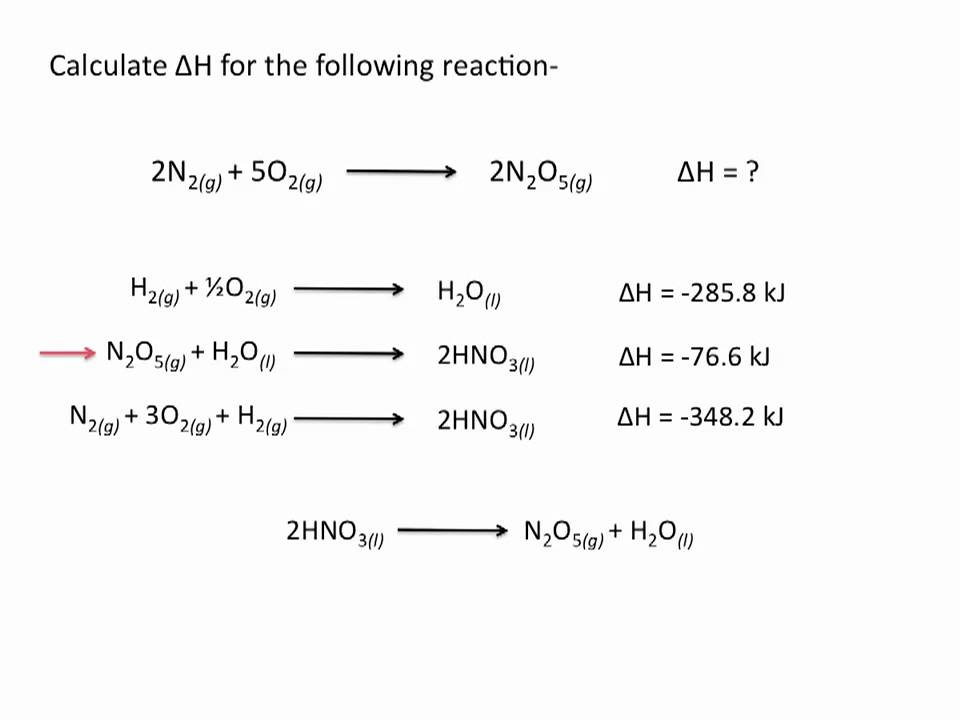  
Determine whether your reaction gains or loses energy. The free energy change of a reaction is a mathematical combination of the enthalpy change and the entropy change.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
